A New Dawn for Genetic Blood Disorders
Cambridge, MA – The medical community is abuzz with excitement as initial results from expanded Phase 3 clinical trials for CRISPR-based gene editing therapies continue to demonstrate remarkable long-term efficacy and safety profiles. These pioneering treatments, primarily focused on genetic blood disorders like sickle cell disease (SCD) and transfusion-dependent beta-thalassemia (TDT), are poised to revolutionize patient care, offering what many hope will be functional cures rather than mere symptom management.
For decades, patients with severe forms of these conditions have faced limited treatment options, often relying on chronic blood transfusions, bone marrow transplants (with their inherent risks and donor matching challenges), or pain management. The advent of CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) technology has opened an entirely new therapeutic avenue, allowing scientists to precisely edit faulty genes responsible for these diseases.
Unprecedented Efficacy and Safety
The latest data from ongoing trials, particularly those involving exagamglogene autotemcel (exa-cel), a therapy developed by Vertex Pharmaceuticals and CRISPR Therapeutics, are particularly compelling. Patients treated with exa-cel for severe SCD have largely achieved sustained freedom from vaso-occlusive crises (VOCs), the hallmark painful episodes of the disease. Similarly, those with TDT have shown sustained independence from blood transfusions. These improvements have been maintained over several years, indicating a durable therapeutic effect.
Safety data has also been robust, with most adverse events being mild to moderate and related to the conditioning regimen (chemotherapy) required before the gene-edited cells are infused, rather than the gene editing itself. This long-term safety profile is crucial for regulatory bodies considering widespread approval. The U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) are currently reviewing these therapies, with decisions anticipated in the coming months.
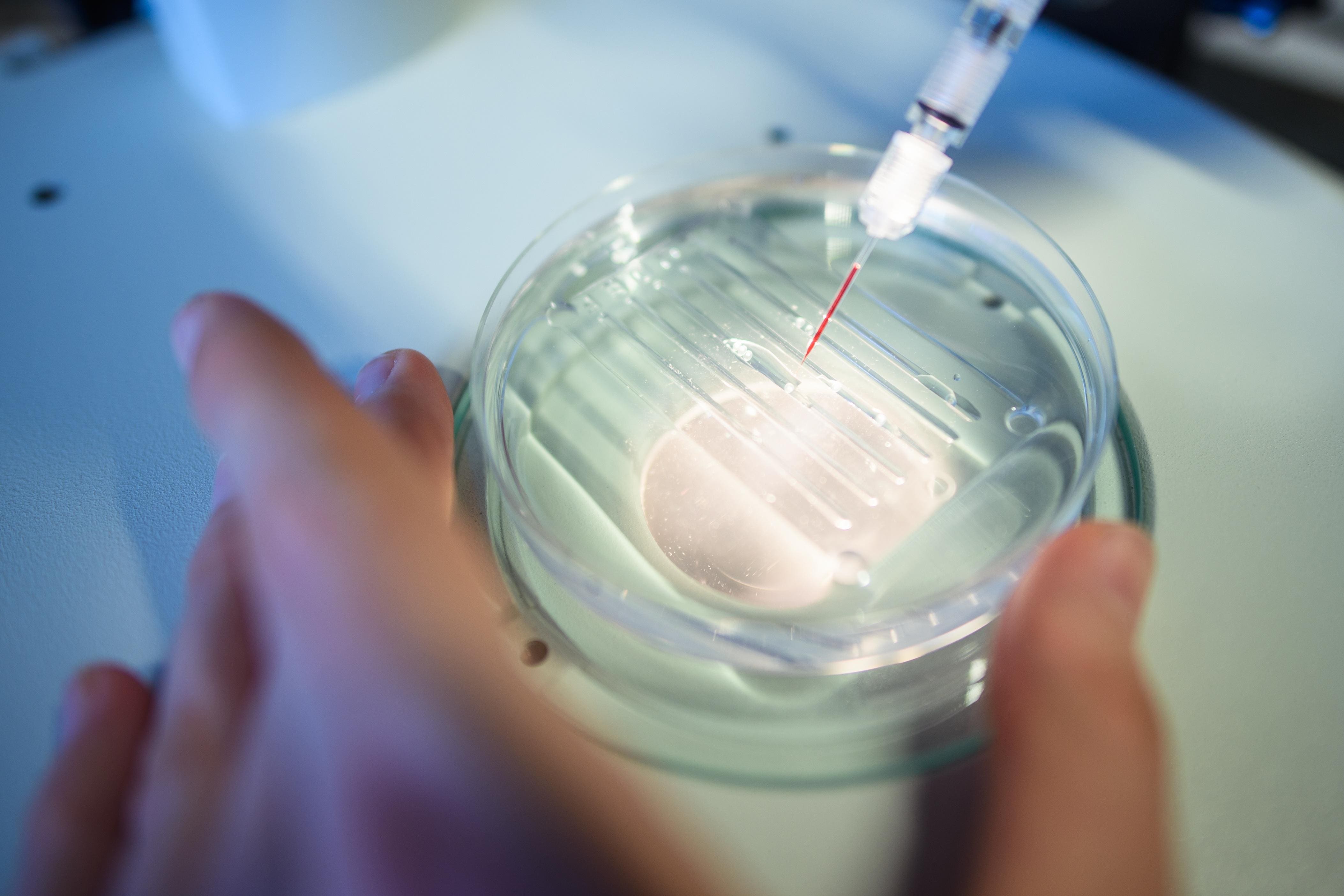
The Mechanism of Action
CRISPR gene editing works by taking a patient's own hematopoietic stem cells, editing them in a laboratory to correct or compensate for the genetic mutation, and then reinfusing them into the patient. For SCD and TDT, the goal is often to reactivate fetal hemoglobin production, which is naturally high at birth but decreases afterward. Fetal hemoglobin is not affected by the genetic mutations causing adult hemoglobin problems, thus alleviating symptoms. This intricate process requires sophisticated biotechnology and rigorous quality control.
This approach represents a significant departure from traditional gene therapy, which often involves introducing new genes using viral vectors. CRISPR's precision offers the potential for fewer off-target effects and a more controlled genetic modification. For more detailed information on the scientific principles behind CRISPR, resources like the Broad Institute of MIT and Harvard provide excellent educational materials: https://www.broadinstitute.org/what-is-crispr.
Anticipation and Future Outlook
The anticipation surrounding these therapies is palpable among patient advocacy groups, clinicians, and investors. If approved, these CRISPR-based treatments could offer a one-time therapeutic intervention, potentially curing patients and dramatically improving their quality of life. This would also significantly reduce the long-term healthcare burden associated with managing chronic genetic disorders.
While the initial cost of these therapies is expected to be high, the long-term benefits in terms of reduced hospitalizations, transfusions, and improved productivity could make them cost-effective over a patient's lifetime. The success of these trials also paves the way for CRISPR technology to be applied to a broader range of genetic diseases, ushering in a new era of genomic medicine. The journey from scientific discovery to clinical reality has been swift and impactful, promising a healthier future for many.
For more information, visit the official website.