A New Era in Genetic Medicine
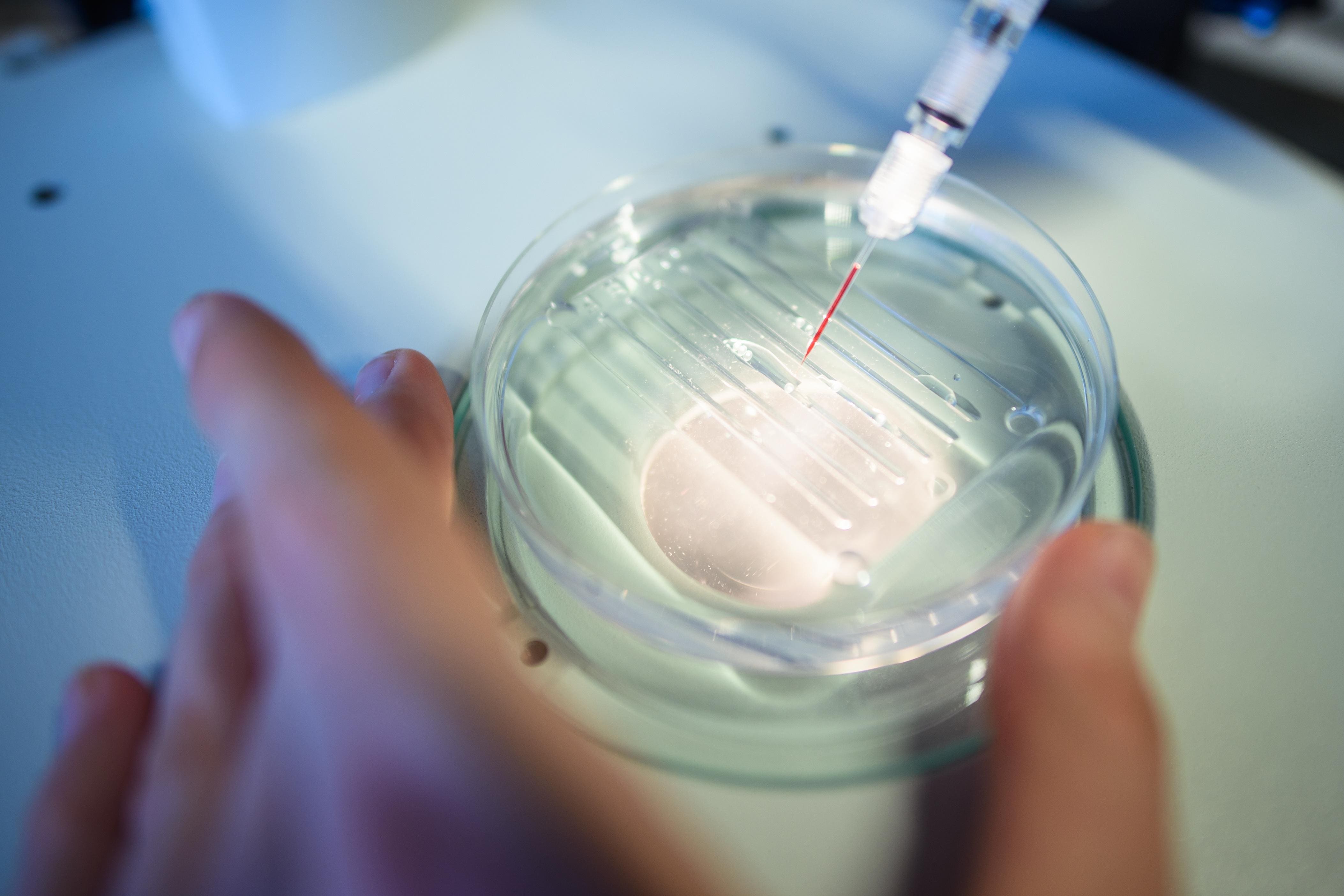
Cambridge, MA – The landscape of genetic medicine is undergoing a profound transformation, driven by groundbreaking advancements in CRISPR gene-editing technology. Recent clinical trial data, unveiled by leading biotechnology firms, highlight the remarkable long-term efficacy of in-vivo CRISPR therapies in directly addressing the root causes of several debilitating inherited diseases. These findings are not merely incremental improvements; they represent a potential paradigm shift in how we approach conditions once considered untreatable.
For years, CRISPR (Clustered Regularly Interspaced Short Palindromic Repeats) has held immense promise, offering a molecular 'cut and paste' tool to precisely modify DNA. The challenge, however, has been safely and effectively delivering this technology directly into a patient's body (in-vivo) and ensuring its sustained therapeutic effect. The latest trial results, focusing on conditions such as certain forms of inherited blindness, severe sickle cell disease, and specific metabolic disorders, indicate that these hurdles are being overcome. Patients in these trials have shown sustained improvements in their conditions, with some exhibiting near-normal physiological function years after a single treatment.
Unprecedented Efficacy and Safety Profiles
The data, presented at a recent international genetics conference, underscore not only the therapeutic power but also the encouraging safety profile of these in-vivo approaches. Researchers reported minimal off-target edits – unintended changes to the DNA – and a low incidence of severe adverse events. This meticulous attention to safety is paramount for any new therapy, especially one that permanently alters a patient's genetic code. The long-term follow-up data, extending beyond two years for several cohorts, provides critical evidence that the benefits are durable, potentially offering a one-time curative treatment rather than ongoing management.
One of the most compelling examples comes from trials targeting Leber Congenital Amaurosis (LCA), a rare genetic eye disease causing severe vision impairment from birth. Patients treated with an in-vivo CRISPR therapy targeting the CEP290 gene have shown significant and sustained improvements in visual acuity and light sensitivity. This success is particularly impactful as LCA previously had no effective treatments, leaving patients to cope with progressive vision loss. Details on these specific trials and the technology can often be found on the websites of companies like Intellia Therapeutics (https://www.intelliatx.com/) or Editas Medicine, pioneers in this field.
Accelerated Review and Accessibility Debates
The impressive outcomes have not gone unnoticed by regulatory bodies. Both the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have indicated an intent to accelerate the review process for these promising therapies. This fast-tracking reflects the urgent unmet medical need for these conditions and the compelling evidence of clinical benefit. While this is excellent news for patients, it also ignites crucial discussions about broader accessibility and equitable distribution.
Gene therapies, by their very nature, are complex and expensive to develop and administer. Ensuring that these potentially life-saving treatments are not limited to a privileged few will be a significant challenge for healthcare systems worldwide. Policymakers, insurers, and patient advocacy groups are already engaging in dialogues about innovative payment models, global manufacturing capabilities, and ethical considerations to ensure that the promise of CRISPR can reach all who need it. As these therapies move closer to approval, the focus will shift from 'can we?' to 'how can we ensure everyone benefits?' – a question that will define the next chapter of genetic medicine.
For more information, visit the official website.