A New Dawn for Genetic Blood Disorders
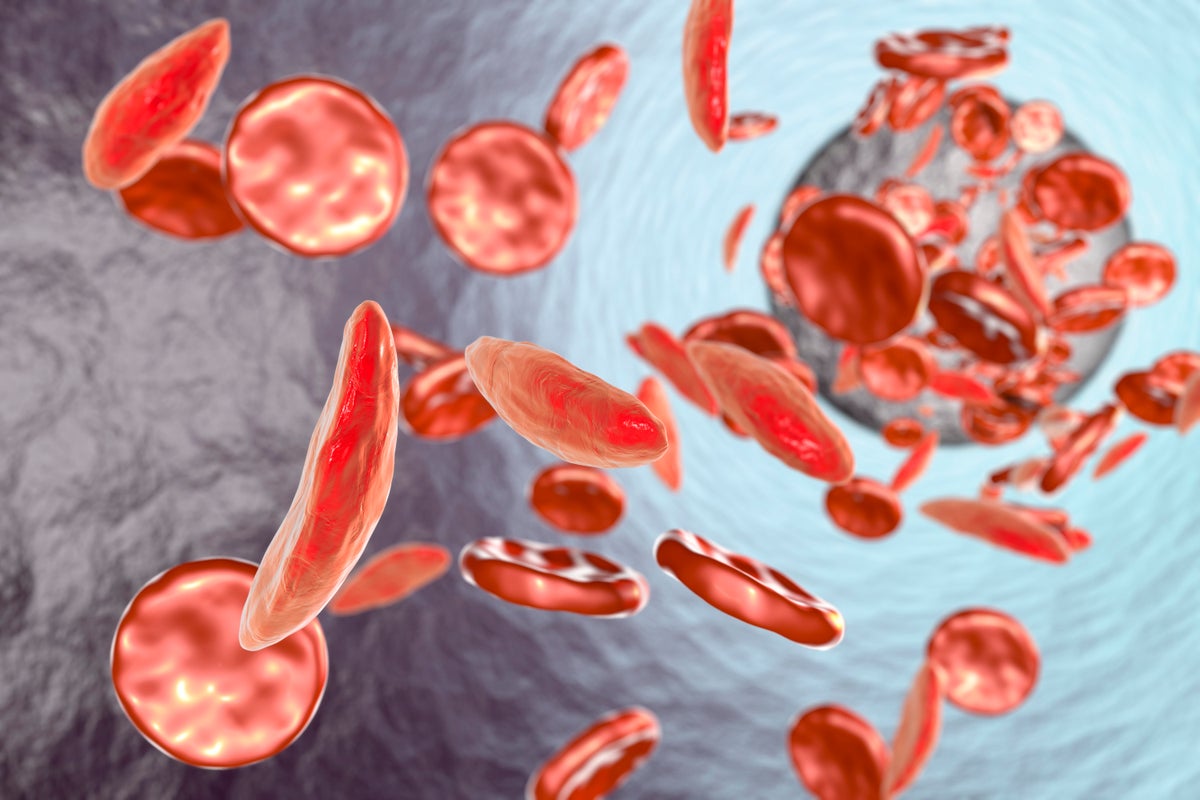
In a significant development for medical science, expanded Phase 2 clinical trials employing CRISPR-Cas9 gene editing have demonstrated remarkable long-term efficacy and an encouraging safety profile in treating severe genetic blood disorders. Patients suffering from sickle cell disease and transfusion-dependent beta-thalassemia, conditions that have historically presented immense challenges for treatment, are now experiencing sustained benefits, offering a beacon of hope for millions worldwide.
The trials, spearheaded by leading biotechnology firms, have focused on an ex vivo approach, where a patient's own hematopoietic stem cells are harvested, genetically modified using CRISPR-Cas9 to correct the underlying genetic defect, and then reinfused. This personalized medicine strategy aims to provide a functional cure by enabling the body to produce healthy red blood cells. Initial reports from these expanded cohorts indicate a dramatic reduction, and in many cases, complete elimination, of debilitating symptoms such as vaso-occlusive crises in sickle cell patients and the need for regular blood transfusions in beta-thalassemia patients.
Unpacking the Science: How CRISPR Works
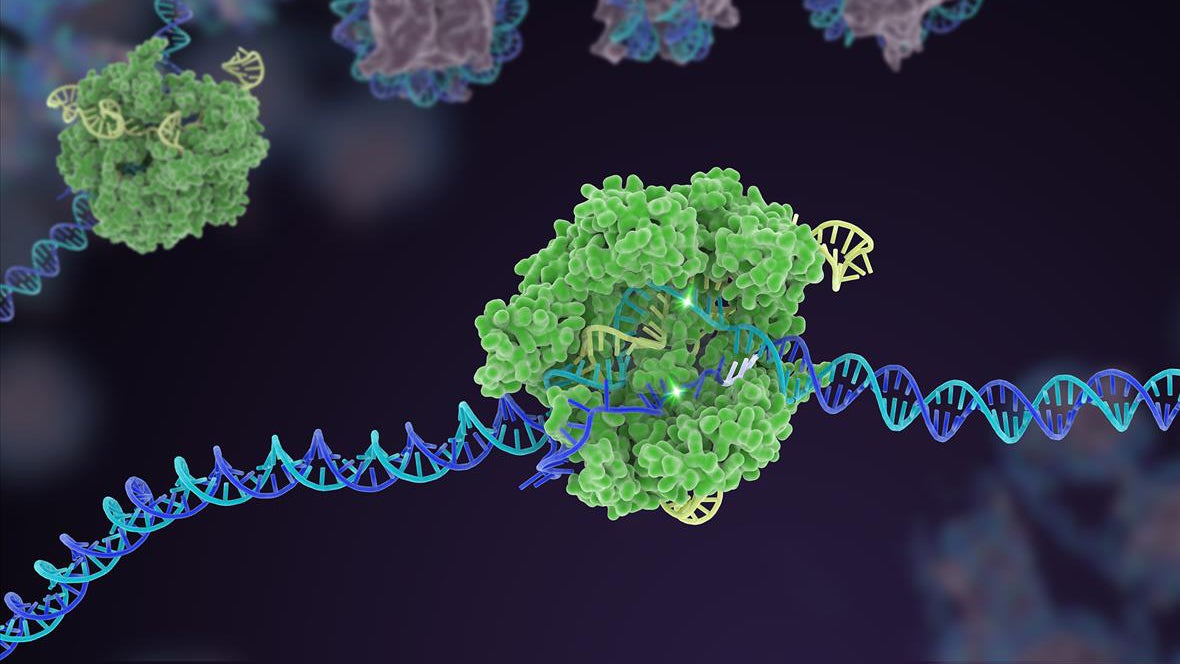
CRISPR-Cas9, often described as 'molecular scissors,' is a revolutionary gene-editing tool that allows scientists to precisely cut and modify DNA sequences. In the context of these trials, the technology is used to either correct the specific mutation responsible for sickle cell disease or to reactivate the production of fetal hemoglobin, which can compensate for the defective adult hemoglobin in both conditions. The precision of CRISPR-Cas9 minimizes off-target edits, a critical factor for long-term safety and efficacy.
The treatment process involves several stages: mobilization and collection of stem cells, ex vivo gene editing, a conditioning regimen to prepare the patient's bone marrow, and finally, the reinfusion of the edited cells. Patients are then closely monitored for engraftment and the production of edited cells. The sustained presence of these edited cells, years after treatment, is a key indicator of the therapy's potential for a lasting cure. For a deeper dive into the mechanics of CRISPR technology, the National Human Genome Research Institute offers comprehensive resources on its official website.
Safety and Efficacy: A Closer Look at the Data
While the excitement surrounding these results is palpable, clinical trials prioritize patient safety above all else. The expanded Phase 2 data have continued to show a manageable safety profile, with side effects primarily related to the conditioning chemotherapy regimen, a standard procedure for stem cell transplants. Importantly, no new or unexpected safety concerns directly attributable to the CRISPR-Cas9 editing itself have been reported in the long-term follow-up of these patients.
Efficacy data are equally compelling. Many patients with sickle cell disease have achieved complete freedom from vaso-occlusive crises, a life-threatening complication, and have reported significant improvements in their quality of life. Similarly, beta-thalassemia patients, who previously relied on frequent, often monthly, blood transfusions, have become transfusion-independent, a transformative outcome that dramatically reduces their disease burden and associated risks. These outcomes are not merely statistical improvements but represent profound changes in individuals' lives.
The Road Ahead: Regulatory Approval and Accessibility
The promising results from these expanded Phase 2 trials bring CRISPR-Cas9 therapies closer to potential regulatory approval. Biotech companies involved are actively engaging with regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) to prepare for potential submissions. If approved, these therapies could mark a paradigm shift in the treatment of genetic blood disorders, moving from chronic management to curative interventions.
However, the journey doesn't end with approval. Ensuring equitable access to these complex and potentially expensive treatments will be a critical challenge. Healthcare systems globally will need to address infrastructure requirements, specialized medical training, and reimbursement models to make these life-changing therapies available to all who could benefit. The scientific community and patient advocacy groups are already working to address these future hurdles, aiming to ensure that this scientific breakthrough translates into real-world impact for patients worldwide. The future of medicine, powered by genetic precision, is rapidly unfolding.
For more information, visit the official website.