A New Horizon in Genetic Medicine
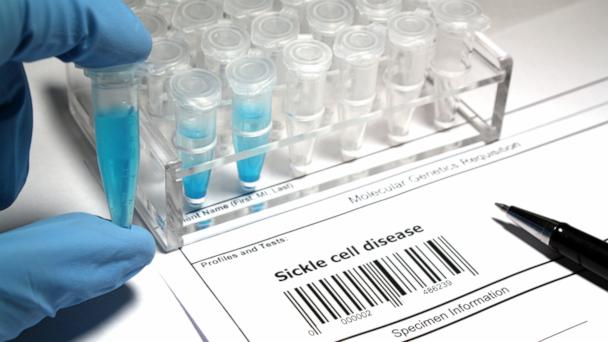
Cambridge, MA – The promise of CRISPR gene editing is rapidly transitioning from laboratory theory to clinical reality, as new data from ongoing clinical trials reveal remarkable success in treating severe genetic blood disorders. Patients suffering from sickle cell disease and transfusion-dependent beta-thalassemia are experiencing life-changing improvements, signaling a potential paradigm shift in how these conditions are managed.
For decades, treatments for genetic blood disorders have largely focused on symptom management, with bone marrow transplants offering a cure but limited by donor availability and significant risks. CRISPR-Cas9, a revolutionary gene-editing tool, offers a more targeted approach: correcting the underlying genetic mutations responsible for these diseases directly within the patient's own cells.
Clinical Trials Yield Promising Results
The latest clinical trial data, presented by companies like Vertex Pharmaceuticals and CRISPR Therapeutics, showcase sustained therapeutic benefits. In trials for exagamglogene autotemcel (exa-cel), a CRISPR-based cell therapy, patients with sickle cell disease have been rendered free of vaso-occlusive crises, the excruciating pain episodes characteristic of the condition. Similarly, patients with beta-thalassemia have achieved transfusion independence, eliminating the need for regular, lifelong blood transfusions.
These results are not merely incremental; they represent a profound improvement in quality of life for participants. The therapy involves extracting a patient's hematopoietic stem cells, editing them ex vivo to correct the genetic defect, and then reinfusing them after a conditioning regimen. While the procedure is intensive, the long-term benefits observed thus far are compelling. Further details on the clinical trials and their methodology can often be found on the National Institutes of Health's clinical trials registry at ClinicalTrials.gov.
The Mechanism of Change
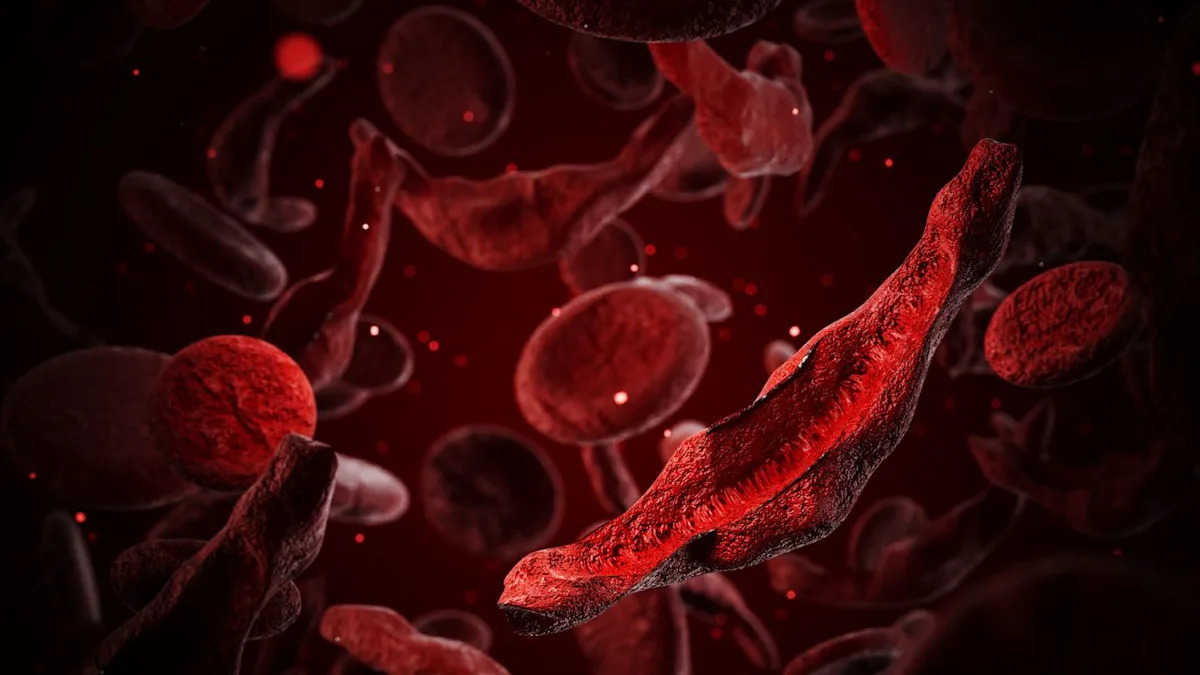
At the heart of this innovation is the CRISPR-Cas9 system, often described as molecular scissors. For sickle cell disease and beta-thalassemia, the therapy targets a specific gene called BCL11A. By editing this gene, the therapy reactivates the production of fetal hemoglobin, a type of hemoglobin naturally produced before birth, which is highly effective at carrying oxygen and does not sickle. This genetic 'switch' effectively compensates for the defective adult hemoglobin, alleviating the disease symptoms.
This precision engineering of the human genome opens doors to treating a myriad of other genetic conditions. Researchers are actively exploring CRISPR applications for cystic fibrosis, Huntington's disease, and certain forms of cancer, among others. The lessons learned from these blood disorder trials are invaluable for refining techniques and ensuring safety for future therapies.
Path to Regulatory Approval and Broader Access
The impressive efficacy and safety profiles observed in these trials are propelling exa-cel towards potential regulatory approval. Both the U.S. Food and Medical Administration (FDA) and the European Medicines Agency (EMA) are currently reviewing the therapy, with decisions anticipated in the coming months. If approved, it would mark a historic milestone, making exa-cel one of the first CRISPR-based therapies available to patients.
While the initial cost of such a sophisticated therapy is expected to be substantial, the long-term benefits of a potential cure could outweigh the lifelong expenses of managing chronic conditions. The broader implications for healthcare systems and patient access will be a significant focus post-approval. The success of these trials underscores the transformative potential of gene editing, promising a future where genetic diseases are not just managed, but cured.
For more information, visit the official website.